 
A single amino acid change in polypeptide chain may change or completely abolish protein function. Sivaranjaniġ5 Remember Every polypeptide chain has a unique amino acid sequence determined by genes. Peptides: di-, three-, tetra- peptides Oligopeptides: up to 20 amino acids Polypeptides: from 20 to 50 amino acids Proteins: > 50 amino acidsįrom “Protein structure” by Dr. Primary structure Definition: The linear sequence of amino acids forming the backbone of a protein. Loss of structure results in loss of biological function.ġ3 The linear sequence of amino acids forming the backbone of a protein. Primary structure Secondary structure Tertiary structure Quaternary structure Native protein structure is essential for the biological function of a protein. Hydrophobic interactions in the formation of protein structure Polar radicals Non-polar radicals PH influences the strength of ion bonds In strong acid: -NH3+ -COOH In strong base: -NH2 -COO-ġ0 Hydrophobic interactions- between hydrophobic (non-polar) radicals Hydrogen bond formation А) between peptide bondsĨ Б) between R-groups of polypeptide chains for example two tyrosine groups or tyrosine and glutamic acid or aspartic acid radicals В) between peptide bond and a side amino acid radical (for example serine, tyrosine)ĩ Ion bond (between oppositely charged radicals) Peptide bond Covalent bonds Disulfide bond Hydrogen bond Non-covalent bonds Ionic bond Hydrophobic interactionsīetween two cysteine molecules Between two cysteine R-groups of polypeptide chains 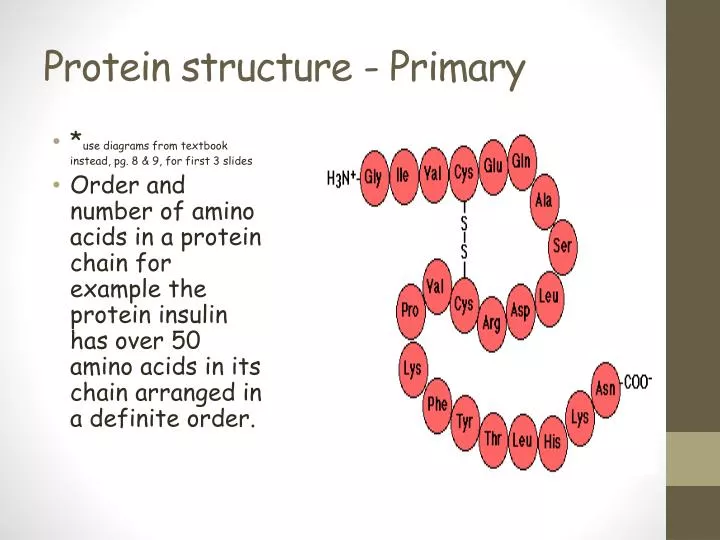
Side chains are free to rotate on either site of the peptide bond. Polypeptide backbone is a repeating sequence of N-C-C-N-C-C… The side chain or R group is not a part of the backbone or the peptide bond.ģ Polypeptide Chain Peptide bond formation:Ĭovalent, strong bond Partial double bond character (distance is 1.32 Å (angstroms) which is midway in a single bond 1.49 Å and a double bond 1.27Å) Rigid and planar Prevailing trans configuration of neighboring α – carbon atoms. Protein structure: primary, secondary, tertiary and quaternary structure.Ģ Proteins are biopolymers, made of the 20 L- α-amino acids linked by peptide bonds. Protein structure: primary, secondary, tertiary and quaternary structure."- Presentation transcript:ġ Lecture 2. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
